The Clinical Data Interchange Standards Consortium (CDISC), FDA and the clinical trial industry are converging on ways to streamline and “…modernize the clinical research system.” With increasing adoption among clinical trials, research sponsors and investigators, clinical trial operations are taking note and heeding more clearly defined standards around collecting data on critical diseases that facilitates regulatory submissions and reporting adverse events
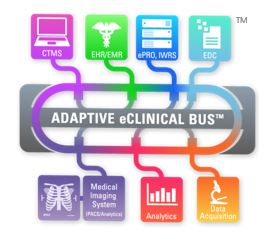
Until then, the use of standard compliant mediated solutions or iPaaS (Adaptive eClinical Bus) is the best approach. It offers a validated, compliant, easy to use and cost effective approach to transferring and sharing data even among disparate systems that originally were not designed to “talk” to one another.
Learn more about how Adaptive Clinical Systems can help improve the interoperability of your clinical trials and help streamline your clinical trial process. Click here to learn more.
Interested in the article, CDISC Standards Key to Meeting FDA Regulations, Research Goals?
Read the full article by By Jill Wechsler:
