Adaptive Clinical Systems, known for its eClinical Bus solution that offers a secure, validated, compliant, and cost-effective solution for clinical data integration, is pleased to announce its partnership with Mint Medical’s mintLesion™ software, making it fully compatible with the latest release – mintLesionTM 3.1.
How does the Adaptive eClinical Bus – mintLesionTM partnership benefit you and your clinical trial operation?
- Seamless data interchange among all system components by study
- Streamlined Clinical trial protocols, criteria, and workflows
- Utilization of your existing technology investments in CTMS and Medical Imaging systems
- Leveraging SaaS Hosting by Adaptive Clinical Systems – delivering accurate, real-time, and validated information
- Study startup time reduced to less than two weeks
- Elimination of data errors due to reentry or manual submission
- Enhanced real-time, on-demand status reporting
- Track record of 100% audit success rate
What is mint LesionTM?
mint Lesion™, by Mint Medical, supports radiologists in performing the radiological Site Read for a clinical trial and provides an optimized workflow that helps to assess the effectiveness of a cancer therapy. The integrated software solutions combine to help maintain regulatory compliance and close oversight and offers increased accuracy and efficiencies in the flow and accessibility of clinical trial data. All data is housed in the strictest compliance with all privacy and security provisions of HIPAA, GxP, and 21 CFR Part-11. Furthermore, detailed audit logs and reports empower DM and QA staff to maintain rigorous oversight of all data at any time.
New Features in mint Lesion™ 3.1
With the new mint LesionTM 3.1, Mint Medical created a simplified and uniform user interface that focuses on the particular context of a current case and gently offers guidance according to the corresponding screening, staging, or response assessment criteria. mint Lesion knows about the intricacies of particular guidelines and automatically determines stage of disease and response to therapy, based on radiological multiparametric image annotations, classifications, and further clinical parameters. The profiles include a variety of tumor-node-metastasis (TNM) tumor entities, staging and transplant scoring of liver disease, screening and staging of prostate carcinoma, and numerous response evaluation criteria (RECIST, mRECIST HCC, Cheson, irRC, irRECIST, Choi, RANO, PCWG2, etc.). In addition, mint Lesion characterizes lesions also by their texture and highlights potential tipping points for clinical treatment decisions during the read process and in the automatically generated structured reports.
What is the Adaptive eClinical Bus®?
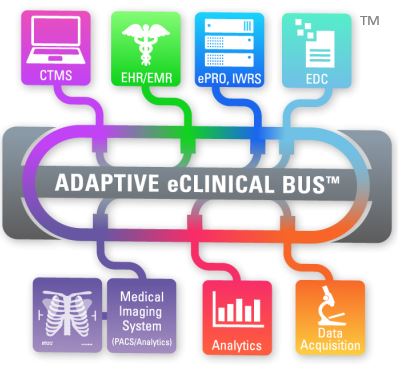
Discover for yourself how the simple integration of the validated Adaptive eClinical Bus® solution with Mint Medical’s solution mint Lesion™ for context-driven read procedures and structured reporting can help your clinical trial operation achieve huge gains in accuracy, efficiency, and interoperability.
Contact Us Today for a Live Demo
Email: [email protected]
Tel: +1 856 473 4370
